Ankles, Arms, Legs
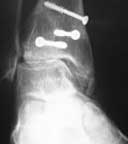
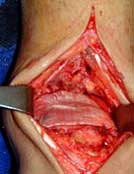
1. Enhancement of Syndesmotic Fusion Rates in Total Ankle Arthroplasty With the Use of Autologous Platelet Concentrate.
Foot & Ankle International. 2005 Jun;26(6):458-61
BACKGROUND: One of the challenges of total ankle arthroplasty continues to be achieving a solid distal fusion of the tibiofibular joint. Delayed union rates of 29% to 38% and the nonunion rates of 9% to 18% for syndesmotic fusion have been documented. The risk of tibial component migration has been reported to increase 8.5 times if a solid syndesmotic fusion is absent. Growth factors have been shown to accelerate bone healing and may enhance the fusion of the syndesmosis and, thereby, decrease the frequency of nonunion and subsequent tibial component migration.
METHODS: An autologous platelet concentrate was used to increase the amount of growth factors at the site of the distal tibiofibular joint fusion in 20 total ankle arthroplasties.
RESULTS: Our 6-month fusion rate was 100%. When compared to historical controls (6-month fusion rate of 62%) the difference was statistically significant (p < 0.0001).
CONCLUSION: The improved rate of distal tibiofibular fusion may be attributable to the increased presence of growth factors provided by an autologous platelet concentrate.
 |
 |
|---|
2. The use of autologous concentrated growth factors to promote syndesmosis fusion in the agility total ankle replacement. A preliminary study.
Foot & Ankle International. 2005 Oct;26(10):840-6
BACKGROUND: The Agility (DePuy, Warsaw, Indiana) total ankle replacement has been in use since 1984. One of the most common complications continues to be delayed union or nonunions of the distal tibiofibular syndesmosis. In the reported studies on the Agility ankle the delayed union and nonunion rate can be as high as 38%. METHODS: Since 1999, 114 Agility total ankle replacements were done at two centers in the United States without the use of autologous concentrated growth factors. Since July of 2001, 66 Agility ankles were implanted with Symphony (DePuy, Warsaw, Indiana) augmented bone grafting. The standard operative technique was followed in all the patients. Prospective data was collected on all patients. The standard ankle radiographs were taken preoperatively and postoperative at 8 weeks, 12 weeks, 16 weeks, 6 months, and yearly. CT scans were obtained at 6 months if fusion at the syndesmosis was questionable. The Graphpad Instat software (Graphpad Software Inc., San Diego, CA) was used for statistical analysis. The two-tailed unpaired t-test was used, and the value <0.05 was considered significant.
RESULTS: There was no statistical difference in the demographic data for the two groups. In 114 ankle replacements without autologous concentrated growth factors 70 fused at 8 weeks (61%), 14 fused at 12 weeks (12%), 13 fused at 6 months (12%). There were 17 nonunions (15%); delayed unions (3 to 6 months) and nonunions, therefore, equaled 27%. The syndesmosis fused in 50 of the 66 ankle replacements (76%) that had autologous concentrated growth fractures at 8 weeks (76%); 12 fused at 3 months (18%), 2 fused at 6 months (3%), 2 had nonunions (3%). Delayed unions (3 to 6 months) and nonunions equaled 6%. There was a statistically significant improvement in the 8- and 12-week fusion rates, and a statistically significant reduction in delayed unions and nonunions.
CONCLUSION: Autologous concentrated growth factors appear to make a significant positive difference in the syndesmosis union rate in total ankle replacements.
3. Evaluation of Bone Healing Enhancement by Lyophilized Bone Grafts Supplemented With Platelet Gel
Journal of biomedical materials research. Part B, Applied biomaterials. 2005 Sep 13
BACKGROUND: Orthopedic practice may be adversely affected by an inadequate bone repair that might compromise the success of surgery. In recent years, new approaches have been sought to improve bone healing by accelerating the rate of new bone formation and the maturation of the matrix. There is currently great interest in procedures involving the use of platelet gel (PG) to improve tissue healing, with satisfactory results both in vitro and in maxillofacial surgery. Otherwise, to our knowledge, only a preliminary clinical study was undertaken in the orthopedic field [Kitoh et al., Bone 2004;35:892-898] and the efficacy of PG is still controversial. Our paper focuses on the effect on bone regeneration by adding PG to lyophilized bone chips used for orthopedic applications.
METHODS: The clinical model and the laboratory methodology were standardized. As a clinical model, we employed the first series of patients of a randomized case-control study undergoing high tibial osteotomy (HTO) for genu varus. Ten subjects were enrolled: in 5 patients lyophilized bone chips supplemented with PG were inserted during tibial osteotomy (group A); 5 patients were used as a control (group B) and lyophilized bone chips without gel were applied. Forty-five days after surgery, computed tomography scan guided biopsies of grafted areas were obtained and the bone maturation was evaluated by a standardized methodology: the osteogenic and angiogenic processes were semi-quantitatively characterized by using histomorphometry, and the mineral component of the lyophilized and host bone was analyzed by using X-ray diffraction technique with sample microfocusing and microradiography.
RESULTS AND CONCLUSION: Lyophilized bone with PG seems to accelerate the healing process, as shown by new vessel formation and deposition of newly formed bone, with no evidence of inflammatory cell infiltrate, when compared with lyophilized bone without gel. On the contrary, lyophilized bone undergoes a resorption process, and a fibrous tissue often fills the spaces between chips. A histiocytic/giant-cell reaction is sometimes present. Otherwise, no differences have been found concerning microstructure. Our findings show the reliability of the methodology used to monitor early bone repair. The completion of the study and the evaluation of the ultimate clinical outcome are necessary in order to verify PG in vivo effects in orthopedic surgery.
 |
|---|
4. Stromal Stem Cells and Platelet-Rich Plasma Improve Bone Allograft Integration
Clinical orthopaedics and related research. 2005 Jun;(435):62-68
BACKGROUND: Early vascular invasion is a key factor in bone allograft incorporation. It may reduce the complications related to slow and incomplete bone integration. Bone-marrow-derived stromal stem cells associated with platelet-rich plasma are potent angiogenic inducers proven to release vascular endothelial growth factor. Our goal was to test whether the combination of stromal stem cells and platelet-rich plasma is able to increase massive allograft integration in a large animal model with sacrifice at 4 months.
METHODS: A critical defect was made in the mid-diaphysis of the metatarsal bone of 10 sheep; the study group received an allograft plus stromal stem cells, platelet-rich plasma, and collagen (six animals) and the control group received only the allograft (four animals). Investigation was done with radiographs, mechanical tests and histomorphometric analysis, including new vascularization.
RESULTS: Results showed substantial new bone formation in the allograft of the study group. Bone formation is correlated with better vascular invasion and remodeling of the graft in the study group.
CONCLUSION: These results confirm the key role played by stromal stem cells and platelet-rich plasma in bone repair. Further studies are needed to better define the role stromal stem cells play when implanted alone.